Of2 Shape And Bond Angle. The actual bond angle for h 2 o is 104.5˚ while the angle for of 2 is 103.1˚. Becl2 has a linear shape with a bond angle of 180 degrees, where as h2o has a bent shape with a bond angle of 104.5 degrees.
Ccl4 is sp3 hybridisation is tetrahedrol shape bond angle is 109º. Of2 the electron geometry is tetrahedral and the molecular shape is bent.the theoretical bond angle is 109.5 °, but repulsions by the lone pairs decrease the bond angle to about 103 °. The bond angle of bef2 is 180º as expected because the beryllium central atom attains sp hybridized and no lone pair present on it.
Hybridization In Chemistry, An Orbit Is A Fixed Path Around The Nucleus In Which Electrons Tend To Move Or Revolve.
This gives scl2 a bond angle of 103°. Ι multiple choice ο trigonal planar, 120 ο bent. The vsepr notation for the of2 molecule is ax2e2.
It Is Because In Case Of O F 2 Electrons Are Nearer To Fluorine Due To High Electronegativity Of F Compared To C L To B R And Because Of This Repulsion Between Electron Decreases;
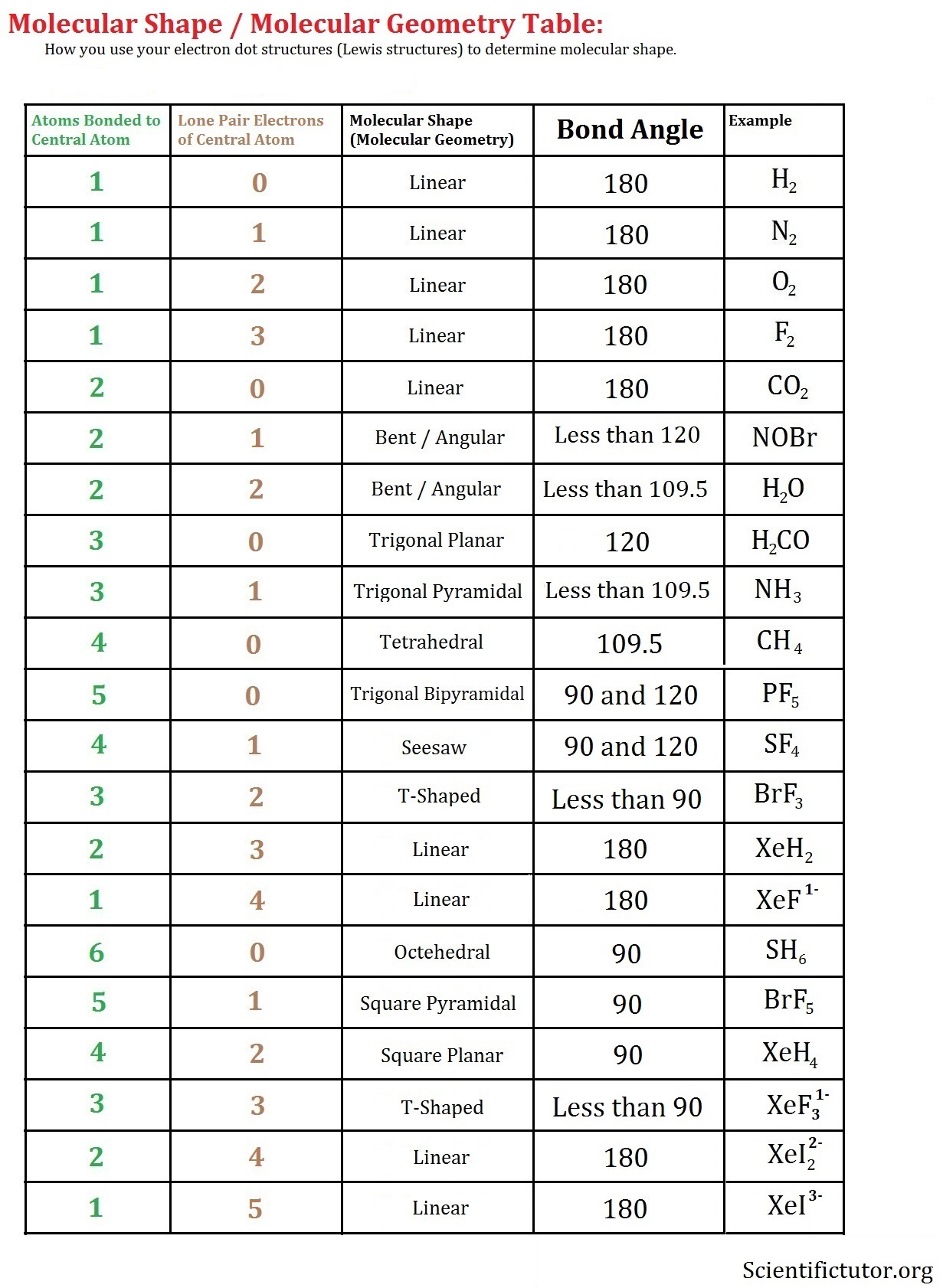
According to the above chart, we have a bent molecule structure. The experimental data shows that the bond angle in the molecules of of 2 and ocl 2 differ from ideal bond angle: I have looked around and most of the sites suggest that bond angle of $\ce{obr2}$ is greater than $\ce{ocl2}$ and it is explained by stating the electronegativity difference and looking at electron repulsion (vsepr).
The Theoretical Bond Angle Is 120 °, But Repulsion By The Lone Pairs Decreases The Bond Angle To About 117 °.
According to the above chart, we have a bent molecule structure. The bond angle is around 103. The middle carbon (c2) has 2 ethyl communities and 2 hydrogen atoms related to it.
The Water Molecule Is Bent Molecular Geometry Because The Lone Electron Pairs, Although Still Exerting Influence On The Shape, Are Invisible When Looking At Molecular Geometry.
Of lone pairs of electrons of the central atom, ‘x = 2. The bond angle is around 103 degrees (due to the repulsion of the lone pair). Predict the molecular shape and give the approximate bond angle in the of2 molecule.
According To The Above Chart, We Have A Bent Molecule Structure.
Of2 the electron geometry is tetrahedral and the molecular shape is bent.the theoretical bond angle is 109.5 °, but repulsions by the lone pairs decrease the bond angle to about 103 °. What is the shape and bond angle for of2? Of lone pairs of electrons of the central atom, ‘x = 2.