Lewis Structure Caffeine. (c) how many valence electrons are used to. “a sigma bond is created by an end to end overlap of atomic orbitals” (nelson chemistry 12).
Every time you are asked to determine the number of pi or sigma bonds in a compound, you must draw its lewis structure. Southwestern supply company has an economic order quantity for item b of 200 units. Caffeine has a total of 25 sigma bonds.
Every Line Represents A Sigma Bond, While Every Double Bond, Represented By Two.
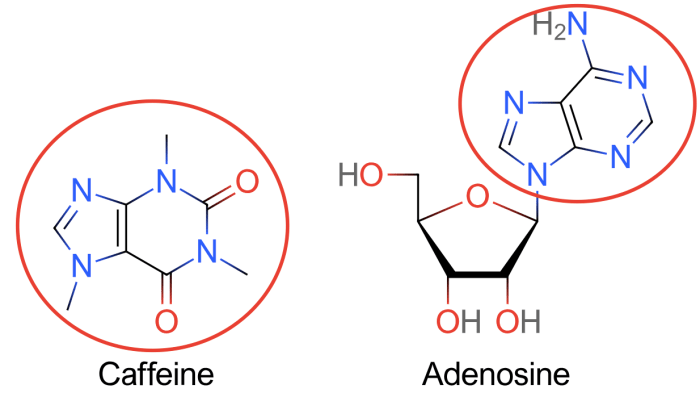
Share to twitter share to facebook share to pinterest. The lewis structure of caffeine is shown below. (c) how many valence electrons are used to.
As Such, The Two Structures Are Note:
In this example, we can draw two lewis structures that are energetically equivalent to each other — that is, they have the same types of bonds, and the same types of formal charges on all of the structures.both structures (2 and 3) must be used to represent the molecule’s structure.the actual molecule is an average of structures 2 and 3, which are called resonance structures. → knowing the shape of a molecule allows us to explain the observed properties and behavior of these substances. “a sigma bond is created by an end to end overlap of atomic orbitals” (nelson chemistry 12).
Free Access To Latest Caffeine Lewis Structure News.
Draw a lewis structure for caffeine in which all atoms have a formal charge of zero. Students also viewed these chemistry questions. We begin with the basic layout of caffeine, merely showing which atoms are bonded together but suppressing any double or triple bonds, as well as any unbonded electrons.
Here's How The Caffeine Molecule Looks Like:
The important thing to remember is that double bonds are comprised of 1 sigma bond and 1 pi bond and triple bonds are comprised of 1 sigma bond and. It may improve weight gain during therapy and reduce the incidence of cerebral palsy as well as reduce language and cognitive delay. Question 168 from chapter eight, which says, consider the following computer generated model of caffeine, which is right here.
Make A Double Bond) Structure.
Ch3 what best describes the geometry for each of the interior… Solution for consider the completed lewis structure for caffeine: How many c and n atoms are sp2 hybridized?